What Chemical Tests Could Be Used to Distinguish Between Acetic Acid and Ethanol
half dozen.4D: Individual Tests
- Page ID
- 95742
Beilstein Exam
The Beilstein test confirms the presence of a halogen in solution, although information technology does not distinguish between chlorine, bromine, or iodine. A copper wire is dipped into the element of group vii-containing solution and thrust into a flame. The copper oxide on the wire reacts with the organic halide to produce a copper-halide compound that gives a blue-greenish color to the flame.
Procedure: In the fume hood, clean a looped copper wire by thrusting information technology into the tip of the bluish cone of a Bunsen burner flame until it glows (Effigy 6.46a). Be sure to "burn off" any residuum liquid on the wire (make sure any greenish flames from previous tests are gone before you brainstorm).
Allow the copper to cool to room temperature, and so dip it into a test tube containing 5-ten drops of your sample, blanket information technology as much every bit possible (Figure half-dozen.46b). If the sample is a solid, adhere some of the solid to the copper wire past first wetting the wire with distilled water so touching information technology to the solid.
Immediately plunge the wire with sample into the blue cone of the flame. A positive issue is a greenish flame, although it might be short-lived and faint (information technology may be easier to see if the fume hood low-cal is turned off). A negative result is the absence of this green color (Figure 6.46c+d).

Benedict'due south Test
The Benedict's test can verify the presence of reducing carbohydrates: compounds that have hemiacetals in their structures and are therefore in equilibrium with the gratis carbonyl class (aldehyde or \(\alpha\)-hydroxyketone). The carbonyl forms are oxidized by the \(\ce{Cu^{2+}}\) in the Benedict'southward reagent (which complexes with citrate ions to preclude the precipitation of \(\ce{Cu(OH)_2}\) and \(\ce{CuCO_3}\)). An insoluble \(\ce{Cu_2O}\) is the inorganic product of this reaction, which usually has a red-brown color (Figure half dozen.47). Carbohydrates with merely acetal linkages are non-reducing sugars and requite a negative result with this test.
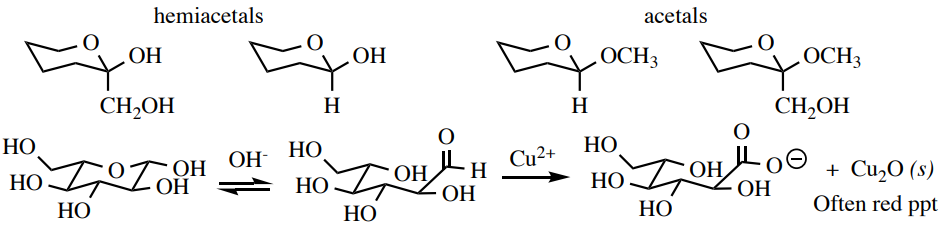
Process: Dissolve \(10\)-\(30 \: \text{mg}\) of solid or 3 drops liquid sample in a minimal amount of h2o \(\left( 0.5 \: \text{mL} \right)\) in a modest test tube (\(13\) ten \(100 \: \text{mm}\)). Add together \(2 \: \text{mL}\) of Benedict'due south reagent.\(^9\) Warm the blue solution in a boiling water bathroom for 2 minutes (Figure vi.48a). A positive consequence is the germination of a cherry-brown solution or precipitate afterwards some fourth dimension, while a negative result is retention of the blue color (Figure 6.48c+d).

Conjugated aldehydes are unreactive in the Bridegroom'southward exam, and the writer constitute many non-conjugated aldehydes to also be unreactive. Formation of colloids seem to prevent the formation of the red precipitate (Effigy six.49 shows the advent of propionaldehyde in the hot water bath, forming a cloudy colloid).

The reaction may only piece of work for compounds that are water soluble (like carbohydrates), as the reaction seems to initiate at the surface (Figure 6.50), and the author establish aldehydes that formed an insoluble layer on the surface to be unreactive.

The Benedict'due south test is related to the Fehling's examination, which uses different ligands on the copper oxidizing species. The Fehling'south reagent uses a \(\ce{Cu^{2+}}\) ion complexed with two tartrate ions.
Bicarbonate Test
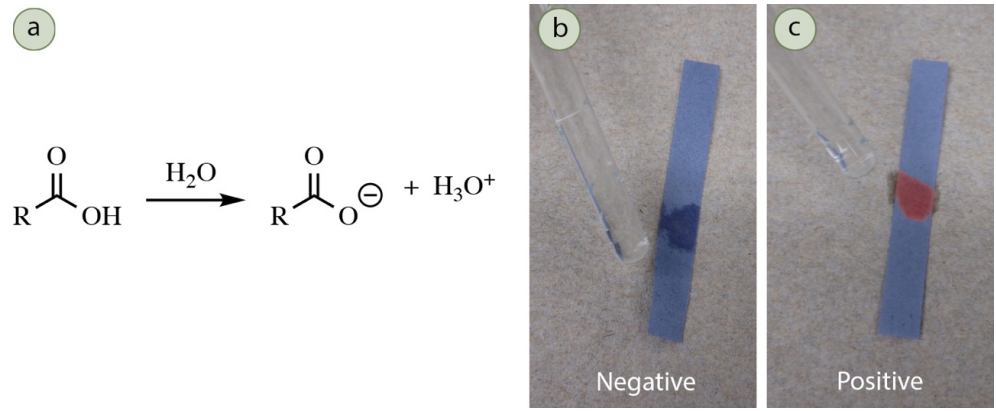
Carboxylic acids and sulfonic acids can react with sodium bicarbonate \(\left( \ce{NaHCO_3} \right)\) to produce carbon dioxide and h2o (Figure 6.51). Other mainstream functional groups (most phenols and alcohols) are not acidic enough to produce a gas with bicarbonate.
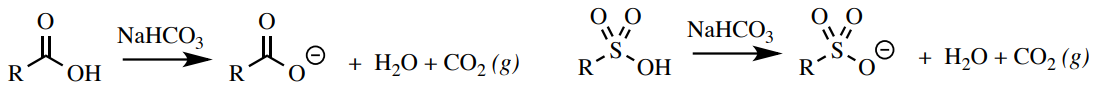
Procedure: Add \(ii \: \text{mL}\) of \(five\% \: \ce{NaHCO_3} \left( aq \right)\) into a test tube and add together 5 drops or \(50 \: \text{mg}\) of your sample. Mix the solution by agitating the test tube. A positive exam for carboxylic acids is the formation of bubbles or frothing (Figure 6.52).

Bromine Test
A solution of bromine in \(\ce{CH_2Cl_2}\) is a test for unsaturation (alkenes and alkynes) and in some cases the power to exist oxidized (aldehydes). The bromine solution is orange and upon reaction the solution turns colorless due to the consumption of bromine. Bromine reacts with alkenes and alkynes through addition reactions and with aldehydes through oxidation (Effigy 6.53). It gives no reaction with aromatics, making this a practiced exam to distinguish alkenes from aromatics.
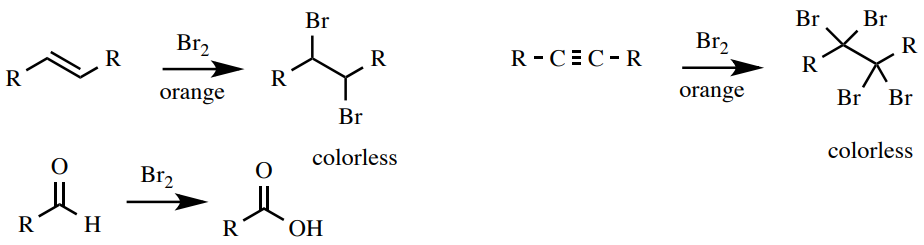
Process: Deliquesce 4 drops or \(50 \: \text{mg}\) of sample in \(ane \: \text{mL}\) of dichloromethane \(\left( \ce{CH_2Cl_2} \correct)\) or i,2-dimethoxyethane. Add together 2 drops of the orange \(5\% \: \ce{Br_2}\) in \(\ce{CH_2Cl_2}\) solution to the exam tube and observe. A positive effect is the immediate disappearance of the orange color to produce a clear or slightly yellow solution (Figure 6.54). A negative consequence is the retentiveness of the orange color. An aldehyde may require a small amount of time to decolorize the solution and produce a positive result (approximately 1 min, Figure half dozen.55) and conjugated aldehydes are unreactive (Figure half dozen.55).


Chromic Acid (Jones) Test
A solution of \(\ce{CrO_3}\) in \(\ce{H_2SO_4}\) is a test for polar functional groups that can be oxidized, which includes aldehydes, main alcohols, and secondary alcohols (Figure vi.57). Tertiary alcohols requite a negative result with this test (Figure 6.56). The orange \(\ce{Cr^{6+}}\) reagent converts to a bluish-green \(\ce{Cr^{three+}}\) species, which often precipitates in acetone.

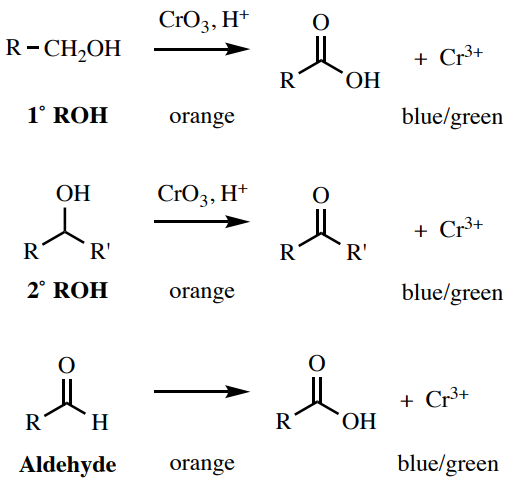
Procedure: Identify \(one \: \text{mL}\) of acetone in a pocket-sized exam tube (\(13\) x \(100 \: \text{mm}\)) and add 2 drops or \(20 \: \text{mg}\) of your sample. While wearing gloves, add 2 drops of the orangish chromic acid reagent\(^{10}\) ( safety note: the reagent is highly toxic!) and mix past agitating. A positive result is a blue-greenish color or dark precipitate, while a negative effect is a yellow-orange solution or precipitate with no dark-colored precipitate (Figure vi.58).
Water works better than acetone to rinse chromium reagents into the waste chalice, although some time needs to be immune for dissolution of the \(\ce{Cr^{3+}}\) species.

2,4-DNPH (Brady's) Test
A solution of two,4-dinitrophenylhydrazine (2,4-DNPH) in ethanol is a test for aldehydes or ketones (Figure 6.59). Most aldehydes or ketones will react with the orange reagent to give a red, orange, or yellow precipitate. Esters and other carbonyl compounds are generally not reactive enough to requite a positive event for this examination.
The colour of the precipitate may give show for the amount of conjugation nowadays in the original carbonyl: an orange precipitate forms for non-conjugated carbonyls (Figure 6.60c shows the result for two-butanone), and a red precipitate forms for conjugated carbonyls (Figure 6.60d shows the result for cinnamaldehyde).
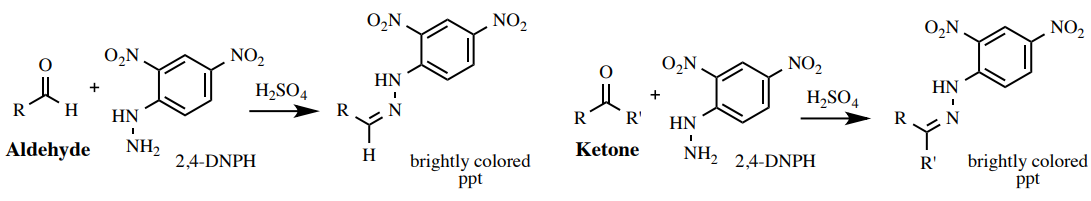
Procedure: Add 3 drops of sample to a minor test tube (\(13\) 10 \(100 \: \text{mm}\)), or dissolve \(10 \: \text{mg}\) of solid sample in a minimal amount of ethanol in the test tube. While wearing gloves, add together about \(ane \: \text{mL}\) of the orange two,iv-DNPH reagent\(^{11}\) ( safety note: the reagent is highly toxic!) and mix the test tube by agitating.
A positive result is the immediate germination of a large amount of brightly colored precipitate (crimson, orange, or yellowish). A negative consequence is the absenteeism of this precipitate and a transparent xanthous-orange solution (Effigy 6.60).

Ferric Hydroxamate Exam
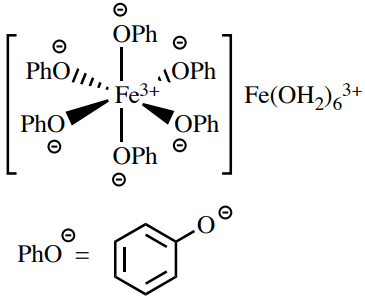
The ferric hydroxamate procedure is a probe for the ester functional group. Esters heated with hydroxylamine produce hydroxamic acids, which form intense, colored complexes (ofttimes dark maroon) with \(\ce{Fe^{three+}}\). A possible structure of these complexes is shown in Figure 6.61. This exam is related to the phenol test, and as in that test, compounds with loftier enolic graphic symbol can give a colored complex with \(\ce{Atomic number 26^{three+}}\). Therefore, a preliminary examination is performed to run into if the carbonyl chemical compound existence tested produces enough enol to form a colored complex with \(\ce{Fe^{3+}}\), which would lead to a imitation positive consequence.
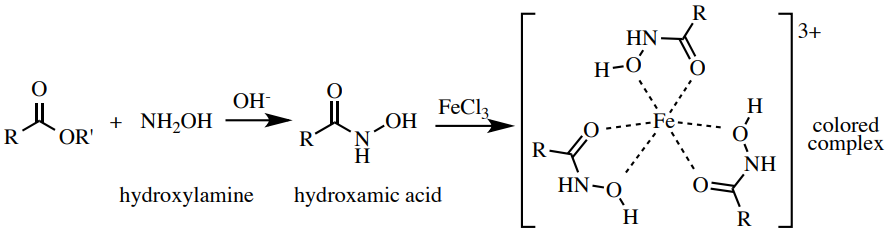
Process: Perform a preliminary test to be certain that this test will not give a false positive. Add the post-obit to a pocket-sized test tube (\(xiii\) x \(100 \: \text{mm}\)): \(one \: \text{mL}\) ethanol, 2 drops or \(20 \: \text{mg}\) of your sample, \(i \: \text{mL}\) of \(i \: \text{G} \: \ce{HCl} \left( aq \correct)\), and 2 drops of \(5\% \: \ce{FeCl_3} \left( aq \right)\) solution. If the solution is clear or yellowish (the color of the \(\ce{FeCl_3}\), Figure 6.62a), this exam will work and non produce a imitation positive (proceed on). If a definite color other than yellow appears, this test will non work for your sample, as it forms a colored circuitous with \(\ce{Fe^{three+}}\) even without hydroxylamine.
Into a clean medium sized test tube (\(18\) 10 \(150 \: \text{mm}\)), add together \(1 \: \text{mL}\) of \(0.five \: \text{M}\) aqueous hydroxylamine hydrochloride \(\left( \ce{NH_2OH} \cdot \ce{HCl} \correct)\), \(0.v \: \text{mL}\) of \(six \: \text{Thou} \: \ce{NaOH} \left( aq \right)\), and v drops or \(fifty \: \text{mg}\) of sample. Heat the mixture in a boiling water bath for about 3 minutes (the volume will reduce by about half, Figure vi.62b).
Apace absurd the solution by immersing it in a tap water bathroom, so add together \(ii \: \text{mL}\) of \(1 \: \text{Grand} \: \ce{HCl} \left( aq \correct)\). If the solution becomes cloudy, add enough ethanol to clarify it. So add 6-10 drops of a yellow \(v\% \: \ce{FeCl_3} \left( aq \right)\) solution. Vigorously mix the tube.
A positive effect is a deep burgundy, umber, or magenta color (ruddy/brown) while a negative upshot is any other color (Figure 6.62c+d). Annotation: apply water to rinse out the test tubes,and if a red consequence won't easily make clean up, add a few drops of \(six \: \text{M} \: \ce{HCl}\).

Iodoform Test
A solution of iodine \(\left( \ce{I_2} \right)\) and iodide \(\left( \ce{I^-} \right)\) in \(\ce{NaOH}\) can be used to exam for methyl ketones or secondary alcohols adjacent to a methyl grouping. This is a very specific test that will requite a positive result (germination of a canary yellow precipitate) but for compounds with the construction \(\ce{RCH(OH)CH_3}\) or \(\ce{RC=OCH_3}\) (Figure 6.63). It does not work for all alcohols or ketones, and does not piece of work well for water-insoluble compounds.
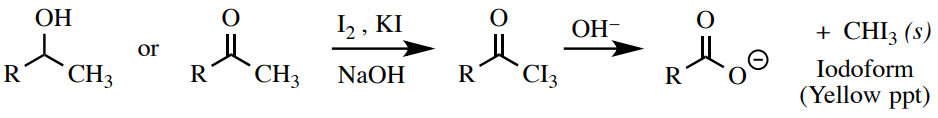
Procedure: Add x drops sample to a small exam tube (\(13\) 10 \(100 \: \text{mm}\)) or \(0.10 \: \text{g}\) dissolved in the minimal amount of one,ii-dimethoxyethane followed past \(1 \: \text{mL}\) of \(10\% \: \ce{NaOH} \left( aq \right)\). Next add ten drops of the dark chocolate-brown iodoform reagent\(^{12}\) (\(\ce{I_2}/\ce{KI}\) solution) and vigorously mix the test tube by agitating.
A positive result is a cloudy yellowish solution, or a yellow precipitate. A negative outcome is a articulate, yellow, or orange solution with no precipitate (Figure half-dozen.64).
If the sample is not water soluble, a pocket-sized organic layer separate from the solution may be seen (it will likely be on top). This layer may become dark xanthous or brown from dissolving the iodine. Vigorously mix the tube to encourage a reaction, but if the darkened organic layer remains and no precipitate forms, this is still a negative result (Figure 6.64d).
Note: a false positive result may occur if the examination tube was cleaned with acetone before utilize, and residual acetone remained in the tube.

Lucas Test
The Lucas reagent (concentrated \(\ce{HCl}\) and \(\ce{ZnCl_2}\)) is a test for some alcohols. Alcohols can react through an \(S_\text{N}1\) mechanism to produce alkyl halides that are insoluble in the aqueous solution and appear every bit a white precipitate or cloudiness. The examination cannot exist used for water-insoluble alcohols (by and large > 5 carbon atoms), as they may produce a cloudiness or 2nd layer regardless if any reaction occurred or non.
\[2^\text{o} \: \text{or} \: three^\text{o} \: \ce{ROH} + \ce{HCl}/\ce{ZnCl_2} \rightarrow \ce{RCl} \left( s \right)\]
Every bit the mechanism is \(S_\text{Due north}1\), a tertiary alcohol should react immediately, a secondary alcohol react more slowly (perhaps in 5 minutes if at all) and primary alcohols oftentimes don't react at all. Benzylic alcohols \(\left( \ce{Ph-C-OH} \right)\), allylic alcohols \(\left( \ce{C=C-C-OH} \right)\) and propargylic alcohols \(\left( \ce{C \equiv C-C-OH} \right)\) often requite immediate results just like tertiary alcohols.
Procedure: Place \(2 \: \text{mL}\) of the Lucas reagent\(^{xiii}\) ( rubber note: the reagent is highly acidic and corrosive!) into a small test tube (\(13\) x \(100 \: \text{mm}\)). Add together 10 drops of sample, and mix by agitating the test tube.
A positive result is a white cloudiness inside 5 minutes or a new organic layer \(\left( \ce{RCl} \right)\) formation on the top.\(^{xiv}\) A negative consequence is the absence of any cloudiness or merely one layer (Figure 6.65).

Permanganate (Baeyer) Exam
A potassium permanganate \(\left( \ce{KMnO_4} \right)\) solution is a test for unsaturation (alkenes and alkynes) or functional groups that can be oxidized (aldehydes and some alcohols, Effigy 6.66). The permanganate ion \(\left( \ce{MnO_4^-} \correct)\) is a deep majestic colour, and upon reduction converts to a dark-brown precipitate \(\left( \ce{MnO_2} \correct)\). Permanganate cannot react with aromatics, and so is a skillful test to discern between alkenes and aromatics. A positive reaction with alcohols is not e'er dependable (a negative outcome is seen with benzyl alcohols in Figure half-dozen.67).
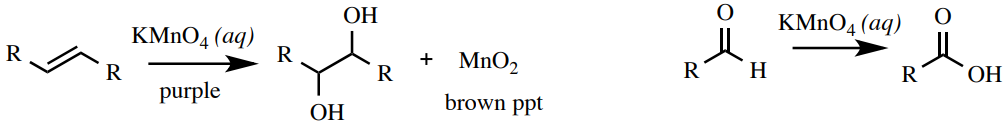
Procedure: Dissolve iv drops or \(40 \: \text{mg}\) of sample in \(1 \: \text{mL}\) of ethanol (or 1,2-dimethoxyethane) in a small-scale test tube (\(xiii\) ten \(100 \: \text{mm}\)). While wearing gloves, add three drops of the deep purple \(1\% \: \ce{KMnO_4} \left( aq \right)\) solution to the examination tube ( rubber notation: reagent is corrosive and will stain skin brown!). Mix the test tube with agitation, and allow it to sit for 1 minute. A positive result is the appearance of a brown color or precipitate. A negative result is a deep purple with no precipitate (unreacted \(\ce{KMnO_4}\), Effigy 6.67).

pH Test
Carboxylic acids and sulfonic acids produce acidic aqueous solutions (Effigy 6.68a), which can be confirmed past turning bluish litmus newspaper pink. The paper changes color (Figure vi.68c) as the indicator molecules react in the lowered pH and form a structure that has a different color.
Procedure: Deliquesce iii drops or \(thirty \: \text{mg}\) of sample in \(1 \: \text{mL}\) of h2o. Dip a glass stirring rod into the solution and touch the rod to blue litmus paper. A positive outcome is a pink or red color on the litmus newspaper (Effigy 6.68c). If the sample doesn't dissolve in water, instead dissolve the same amount of unknown in \(1 \: \text{mL}\) of ethanol. Add together enough water to make the solution barely cloudy. Then add a few drops of ethanol to turn the solution articulate again, and test with the litmus newspaper.
Phenol Test
A ferric chloride solution is a test for phenols, every bit they form intensely colored complexes with \(\ce{Fe^{iii+}}\) (often night blueish). The actual structure of these complexes is debated,\(^{xv}\) simply may be of the general form in Effigy six.69. Some carbonyl compounds with high enol content tin can give false positives with this test.
Process: Place \(1 \: \text{mL}\) water in a small examination tube (\(13\) x \(100 \: \text{mm}\)) forth with either 3 drops or \(thirty \: \text{mg}\) of sample. Add 3 drops of the xanthous \(v\% \: \ce{FeCl_3} \left( aq \correct)\) solution, and mix past agitating.
A positive result is an intense blue, purple, red, or light-green colour while a negative effect is a yellow color (the original color of the \(\ce{FeCl_3}\) solution, Figure 6.seventy).

Silver Nitrate Test
A dilute solution of silver nitrate in ethanol is a test for some alkyl halides. Silverish has a high affinity for halogens (forms strong \(\ce{AgX}\) ionic bonds), and so encourages an \(S_\text{N}ane\) machinery. For this reason, 3rd alkyl halides react faster than secondary alkyl halides (which may or may not react, even with heating), and main alkyl halides or effluvious halides give no reaction. Benzylic \(\left( \ce{PhCH_2X} \right)\) and allylic \(\left( \ce{CH_2=CHCH_2X} \right)\) alkyl halides volition also requite a fast reaction. A positive test result is the germination of the insoluble \(\ce{AgX}\) (Figure half-dozen.71). \(\ce{AgCl}\) and \(\ce{AgBr}\) are white solids, while \(\ce{AgI}\) is a yellow solid.
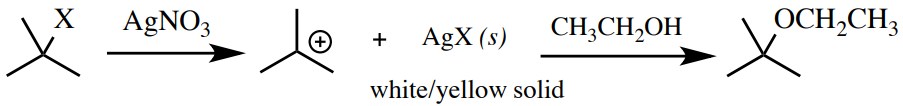
Procedure: In a small test tube (\(13\) ten \(100 \: \text{mm}\)), add \(2 \: \text{mL}\) of \(1\% \: \ce{AgNO_3}\) in ethanol solution. Add four drops of liquid sample or \(40 \: \text{mg}\) fo solid dissolved in the minimal amount of ethanol. Mix the test tube past agitating. Some compounds will have an initial insolubility when first mixed, but the solid oft dissolves with swirling. A positive event is a sustaining white or yellow cloudiness. If cloudiness does not occur within 5 minutes, estrus the tube in a \(100^\text{o} \text{C}\) water bathroom for 1 minute (Effigy 6.72b). Absence of cloudiness even at \(100^\text{o} \text{C}\) is a negative result (Figures vi.72+6.73).

For reactions that produce an intense precipitate, the solution may besides plough blueish litmus paper pink (Figure six.73c+d). An analysis of the reaction mechanism tin can explicate the source of this acidity.

Sodium Iodide (Finkelstein) Test
A solution of sodium iodide in acetone is a test for some alkyl chlorides and bromides. The mechanism is largely \(S_\text{Northward}ii\), and so primary alkyl halides react faster than secondary alkyl halides, and tertiary alkyl halides mostly give no reaction. The reaction is driven by the precipitation of the \(\ce{NaCl}\) or \(\ce{NaBr}\) in the acetone solvent. Therefore, a positive test consequence is the appearance of a white cloudiness (\(\ce{NaX}\) solid).
\[\begin{assortment}{ccccccccc} \ce{CH_3CH_2X} & + & \ce{NaI} \: \text{(acetone)} & \rightarrow & \ce{CH_3CH_2I} & + & \ce{NaX} \left( s \right) & & \left( \ce{X} = \ce{Cl}, \ce{Br} \right) \\ & & & & & & \text{white solid} & & \end{array}\]
Process: In a small test tube (\(xiii\) ten \(100 \: \text{mm}\)), add \(two \: \text{mL}\) of \(xv\% \: \ce{NaI}\) in acetone solution.\(^{16}\) Add 4 drops of liquid sample or \(40 \: \text{mg}\) of solid dissolved in the minimal amount of ethanol. Mix the test tube past agitating.
A positive result is a sustaining white cloudiness. If cloudiness does not occur within 5 minutes, heat the tube in a \(fifty^\text{o} \text{C}\) h2o bathroom for 1 minute. Absence of cloudiness fifty-fifty at \(l^\text{o} \text{C}\) is a negative reaction (Figures 6.74+6.75).


Tollens Test
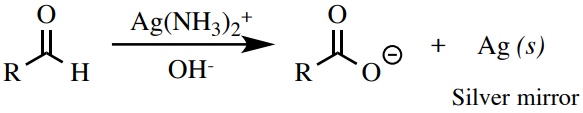
The Tollens reagent \(\left( \ce{Ag(NH_3)_2^+} \right)\) is a balmy oxidizing amanuensis that can oxidize aldehydes, but not alcohols or other carbonyl compounds. A positive test effect is the formation of elemental silver (Figure 6.76), which precipitates out as a "argent mirror" on the examination tube, or every bit a blackness colloidal precipitate.
Procedure: While wearing gloves, mix \(ane \: \text{mL}\) of \(5\% \: \ce{AgNO_3} \left( aq \correct)\) ( safe annotation: toxic!) with \(ane \: \text{mL}\) of \(10\% \: \ce{NaOH} \left( aq \right)\) in a medium sized examination tube (\(18\) ten \(150 \: \text{mm}\)). A dark precipitate of silver oxide will grade (Figure 6.77b). Add dropwise plenty \(ten\% \: \ce{NH_4OH} \left( aq \right)\) to only dissolve the precipitate (notation some time should be allowed between additions). This solution is now the Tollens reagent \(\ce{Ag(NH_3)_2^+}\) (Figure half dozen.77c).
Deliquesce three drops or \(thirty \: \text{mg}\) of sample in a few drops of diethyl ether (omit solvent if compound is water soluble). Add this solution to the \(two\)-\(three \: \text{mL}\) of previously prepared Tollens reagent. Mix the test tubes by agitating. A positive result is a silver mirror on the edges of the exam tube, or formation of a black precipitate. A negative upshot is a clear solution (Figures 6.77d+6.78).
Clean-up: The reagent may grade a very explosive substance (silverish animadvert) over time, and then the examination should be immediately cleaned upward. Acidify the solution with \(5\% \: \ce{HCl} \left( aq \correct)\), then dispose in a waste chalice. A silver mirror can be removed from the glassware past adding a small amount of \(6 \: \text{M} \: \ce{HNO_3} \left( aq \correct)\).


\(^9\)The Benedict'south reagent is prepared as follows, as published past the Flinn Scientific catalog: \(173 \: \text{chiliad}\) of hydrated sodium citrate and \(100 \: \text{g}\) of anhydrous sodium carbonate is added to \(800 \: \text{mL}\) of distilled water with heating. The mixture is filtered, then combined with a solution of \(17.three \: \text{g}\) copper(2) sulfate pentahydrate dissolved in \(100 \: \text{mL}\) distilled water. The combined solutions are diluted to \(1 \: \text{L}\).
\(^{10}\)The chromic acid reagent is prepared as follows: \(25.0 \: \text{thousand}\) of chromium(Half dozen) oxide is added to \(25 \: \text{mL}\) concentrated sulfuric acid, which is then added in portions to \(75 \: \text{mL}\) of water. The reagent has a very long shelf life (10+ years).
\(^{11}\)Training of the two,4-DNPH reagent, as published in B. Ruekberg, J. Chem. Ed., 2005, 82(9), p. A1310, is as follows: To a dry \(125 \: \text{mL}\) Erlenmeyer flask is added \(3 \: \text{g}\) two,four-dinitrophenylhydrazine, \(20 \: \text{mL}\) water and \(70 \: \text{mL}\) of \(95\%\) ethanol. The solution is cooled in an ice bath with stirring, and when at \(x^\text{o} \text{C}\), \(15 \: \text{mL}\) of concentrated sulfuric acid is added slowly in portions. If the temperature exceeds \(xx^\text{o} \text{C}\) during the add-on, the solution should be allowed to absurd to \(x^\text{o} \text{C}\) earlier continuing. The solution is and so warmed to \(60^\text{o} \text{C}\) with stirring, and if solids remain, they are filtered. Finally, the solution is cooled.
\(^{12}\)Preparation of the iodoform reagent is equally follows: \(10 \: \text{g} \: \ce{KI}\) and \(five \: \text{chiliad} \: \ce{I_2}\) is dissolved in \(100 \: \text{mL}\) water.
\(^{13}\)Grooming of the Lucas reagent is as follows: \(160 \: \text{g}\) of fresh anhydrous \(\ce{ZnCl_2}\) is dissolved in \(100 \: \text{mL}\) of cold concentrated \(\ce{HCl}\).
\(^{xiv}\)Although chlorinated organics are typically denser than water, the Lucas reagent has a high quantity of solute, and chlorinated compounds tend to be less dense than the reagent.
\(^{fifteen}\)See Nature, 24 June 1950, 165, 1012.
\(^{16}\)This solution often has a yellow tin to information technology.
Contributor
-
Lisa Nichols (Butte Customs College).Organic Chemistry Laboratory Techniques is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. Complete text is available online.
Source: https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Book:_Organic_Chemistry_Lab_Techniques_%28Nichols%29/06:_Miscellaneous_Techniques/6.04:_Chemical_Tests/6.4D:_Individual_Tests
Post a Comment for "What Chemical Tests Could Be Used to Distinguish Between Acetic Acid and Ethanol"